Matter is anything that has definite composition and property like mass and volume.
A. Level of Organization
Atom -----------Molecule ------------ Element ----------- Compound
Atoms are the building blocks of matter. When bound chemically together they become molecules. An element is composed of one kind of atom. Elements when combined form a compound.
B. Classification according to Physical State
According to physical state matter can be classified as solid, liquid, gas, and plasma.
A. Level of Organization
Atom -----------Molecule ------------ Element ----------- Compound
Atoms are the building blocks of matter. When bound chemically together they become molecules. An element is composed of one kind of atom. Elements when combined form a compound.
B. Classification according to Physical State
According to physical state matter can be classified as solid, liquid, gas, and plasma.
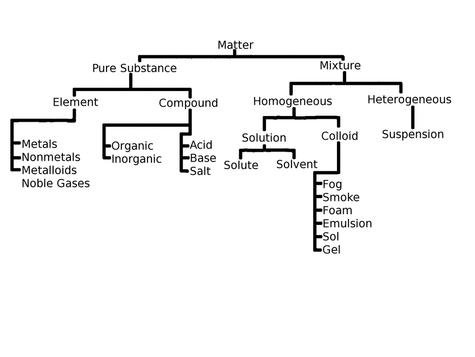
PURE SUBSTANCE
A pure substance is indivisible. It has distinct composition and property.
A pure substance is divided into element and mixture.
An ELEMENT cannot be decomposed into smaller substance by any chemical means. There are four types of elements: metals; nonmetals; metalloids, and noble gases.
COMPOUNDS are chemically bound in a fixed proportion. They can be decomposed into its simpler forms by chemical processes. Compounds can be ORGANIC or INORGANIC.
Compounds can also be classified as acid, base, and salt.
MIXTURE
Mixtures can be broken down to simpler substances by physical means.
There are two types of mixtures: homogeneous and heterogeneous.
HOMOGENEOUS mixtures have uniform composition and show only one phase.
This mixture can either be a solution or a colloid.
HETEROGENEOUS mixtures have no uniform composition and can have two or more phases. SUSPENSIONS are classified under heterogeneous mixtures.
A pure substance is indivisible. It has distinct composition and property.
A pure substance is divided into element and mixture.
An ELEMENT cannot be decomposed into smaller substance by any chemical means. There are four types of elements: metals; nonmetals; metalloids, and noble gases.
COMPOUNDS are chemically bound in a fixed proportion. They can be decomposed into its simpler forms by chemical processes. Compounds can be ORGANIC or INORGANIC.
Compounds can also be classified as acid, base, and salt.
MIXTURE
Mixtures can be broken down to simpler substances by physical means.
There are two types of mixtures: homogeneous and heterogeneous.
HOMOGENEOUS mixtures have uniform composition and show only one phase.
This mixture can either be a solution or a colloid.
HETEROGENEOUS mixtures have no uniform composition and can have two or more phases. SUSPENSIONS are classified under heterogeneous mixtures.
 RSS Feed
RSS Feed